|
12/18/2023 0 Comments Benzophenone ir spectrum 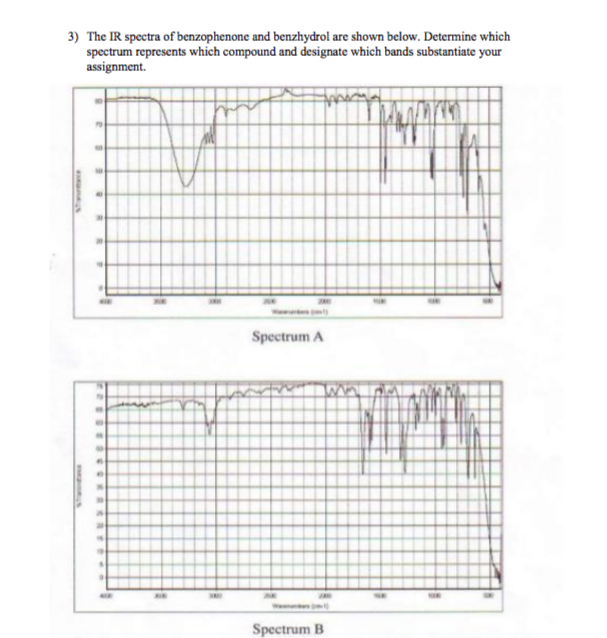
Benzophenone undergoes a rapid intersystem crossing, and therefore quickly decays This is done without the electronĬhanging its spin state, resulting in the molecule existing in singlet excited states S 2 (π,π*) State electrons on its oxygen jump to an antibonding π orbital. 
With the correct wavelength, a benzophenone molecule has one of two paired ground Isopropyl however produces a colorless product. However this results in a slower reaction rate of the benzopinacol and results in a yellow product. Other solvents such as ethanol can be used, Known as the father of photochemistry, Ciamician had left a mixture of isopropylĪlcohol and benzophenone on a window cell. Photoreduction was first discovered in 1900 byĬiamician (1). The synthesis of benzopinacolone was achieved through a two-step synthesis of photoreductionĪnd pinacol-pinacolone rearrangement. ☌ 1 H NMR confirmed the formation of the INTRODUCTION This was found to yieldĩ5% with a melting point of 178-179. Benzopinacol went through a rearrangement to ☌ formīenzopinacolone in glacial acetic acid and using Iodine as a catalyst. Utilizing 2-propanol and a drop of glac In a two-step synthesis, benzopinacol and benzopinacolone were synthesizedialĪcid, benzophenone is photoreduced to benzopinacol. Rapid City, SD 57701, United Supporting InformationĪBSTRACT:through photoreduction and pinacol rearrangement. Synthesis of Benzopinacol and Benzopinacoloneĭepartment of Chemistry and Applied Biological Sciences, South Dakota School of Mines, 501 E. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
